News
06-09-2021
Green Chemistry publication: Furoic acid and derivatives as atypical dienes in Diels–Alder reactions
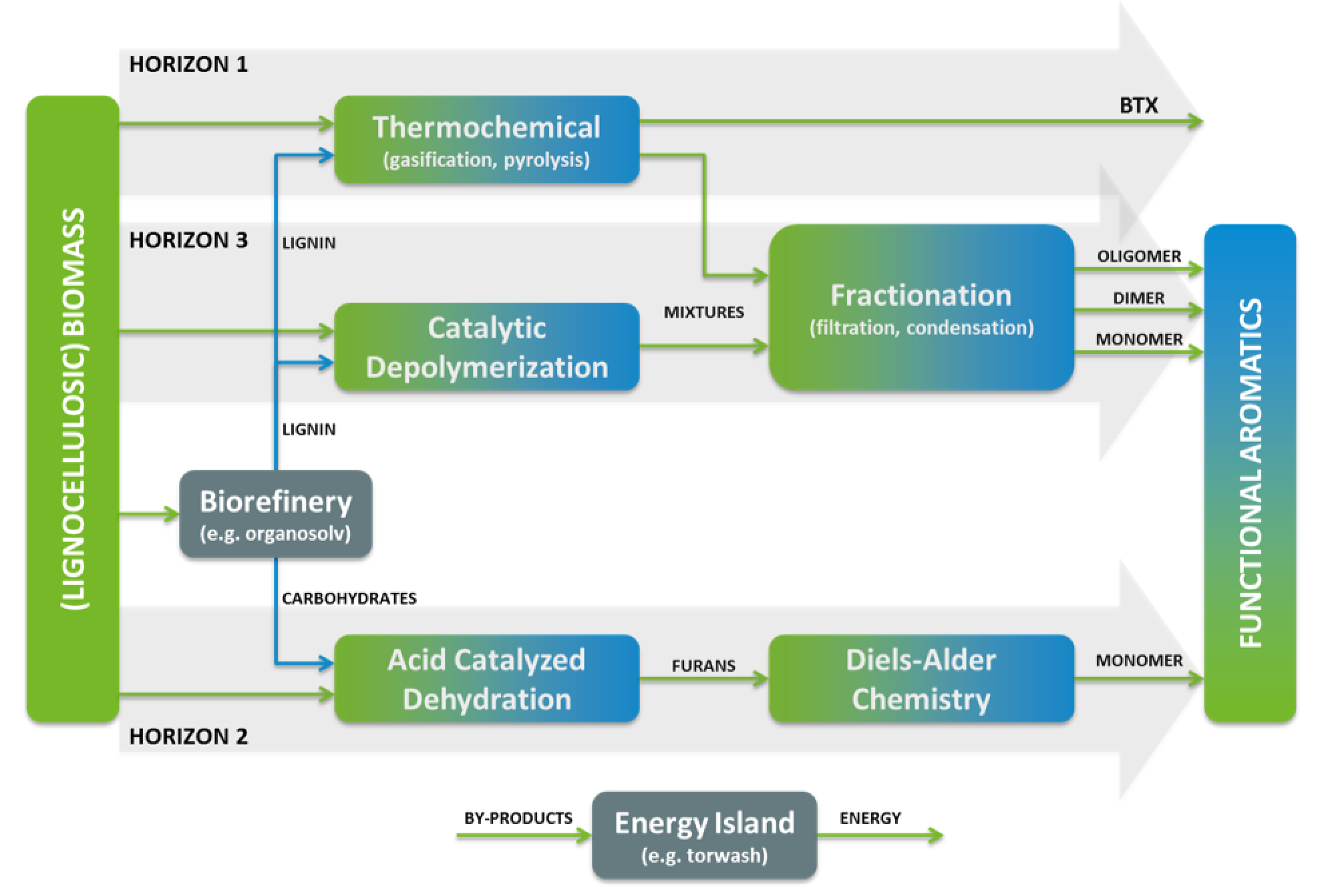
We are proud that - once again – a collaborative research effort involving Biorizon’s lead scientists Marc Crockatt and Dr. Jan Kees van der Waal was recently published in the leading scientific journal Green Chemistry. The Bruijnincx group at Utrecht University and Biorizon co-initiator TNO have developed an efficient furan Diels–Alder cycloaddition reaction, which has the potential to become an important tool in green chemistry, being central to the sustainable synthesis of many chemical building blocks.
Enabling Diels–Alder reactions under very mild conditions
The restriction to electron-rich furans is a significant limitation of the scope of suitable dienes, in particular hampering the use of the furans most readily obtained from biomass, furfurals and their oxidized variants, furoic acids. Herein, it is shown that despite their electron-withdrawing substituents, 2-furoic acids and derivatives (esters, amides) are in fact reactive dienes in Diels–Alder couplings with maleimide dienophiles.
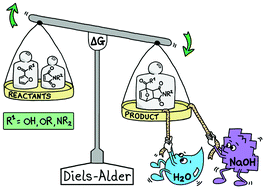
The reactions benefit from a substantial rate-enhancement when water is used as solvent, and from activation of the 2-furoic acids by conversion to the corresponding carboxylate salts. This approach enables Diels–Alder reactions to be performed under very mild conditions, even with highly unreactive dienes such as 2,5-furandicarboxylic acid. The obtained DA adducts of furoic acids are shown to be versatile synthons, providing access to various saturated and aromatic carbocyclic products.
Please read the entire publication in Green Chemistry of the Royal Society of Chemistry (impact factor 9.480) or take a look at the cartoon drawing at the journal's cover that visualizes this research.
Contact
If you want to know more about this research, please contact lead scientist Marc Crockatt at marc.crockatt@tno.nl or +31 (0)6 46 84 74 02.